 |
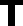 he Council for Responsible Nutri-tion
(CRN) is releasing a new publication, the CRN Reference on Evaluating Botanicals.
The report was compiled for CRN by Marilyn Barrett, Ph.D., a pharmacognosist
and consultant on botanical issues. Barrett said the purpose of the report
is "to give manufacturers, government officials, health care providers,
the media and consumers some perspectives on evaluating botanical products
marketed as dietary supplements." he Council for Responsible Nutri-tion
(CRN) is releasing a new publication, the CRN Reference on Evaluating Botanicals.
The report was compiled for CRN by Marilyn Barrett, Ph.D., a pharmacognosist
and consultant on botanical issues. Barrett said the purpose of the report
is "to give manufacturers, government officials, health care providers,
the media and consumers some perspectives on evaluating botanical products
marketed as dietary supplements."
 For the first time anywhere, the
new CRN publication provides an extensive table showing which of more than
340 botanicals have been monographed in key authoritative references. These
include pharma-copeias such as the US Pharmacopeia-National Formulary, the
British Herbal Pharmacopeia and the American Herbal Pharmacopeia, and other
compilations such as the German Commission E monographs, the World Health
Organization model monographs, and the monographs prepared by the European
Scientific Cooperative on Phytotherapy (ESCOP). Botanicals that are currently
monographed or planned for inclusion in five or more of these authoritative
references include garlic, ginkgo, ginger, ginseng, lemon balm, milk thistle,
peppermint, saw palmetto, St. John's wort, uva-ursi and valerian. For the first time anywhere, the
new CRN publication provides an extensive table showing which of more than
340 botanicals have been monographed in key authoritative references. These
include pharma-copeias such as the US Pharmacopeia-National Formulary, the
British Herbal Pharmacopeia and the American Herbal Pharmacopeia, and other
compilations such as the German Commission E monographs, the World Health
Organization model monographs, and the monographs prepared by the European
Scientific Cooperative on Phytotherapy (ESCOP). Botanicals that are currently
monographed or planned for inclusion in five or more of these authoritative
references include garlic, ginkgo, ginger, ginseng, lemon balm, milk thistle,
peppermint, saw palmetto, St. John's wort, uva-ursi and valerian.
 The CRN report also provides basic
background information on topics such as the past and present use of botanicals,
regulatory status, methodologies for plant identification, approaches to
assessing health benefits and determining dosage, safety evaluation and
standardization. The CRN report also provides basic
background information on topics such as the past and present use of botanicals,
regulatory status, methodologies for plant identification, approaches to
assessing health benefits and determining dosage, safety evaluation and
standardization.
 John Cardellina, Ph.D., a natural
products chemist who joined CRN in April of this year as Director of Botanical
Science and Regulatory Affairs, said, "I view this as one of CRN's
first steps in a campaign to increase awareness and understanding of the
uses, benefits, and safety of botanicals. The report provides a valuable
reference and is an indication of CRN's increased commitment to the botanical
arena." John Cardellina, Ph.D., a natural
products chemist who joined CRN in April of this year as Director of Botanical
Science and Regulatory Affairs, said, "I view this as one of CRN's
first steps in a campaign to increase awareness and understanding of the
uses, benefits, and safety of botanicals. The report provides a valuable
reference and is an indication of CRN's increased commitment to the botanical
arena."
 Cardellina has been a researcher
in natural products for more than 20 years. Before coming to CRN, he was
a senior investigator and Head of the Natural Products Chemistry Section,
Laboratory of Drug Discovery Research and Development, Developmental Therapeutics
Program, National Cancer Institute. Cardellina has been a researcher
in natural products for more than 20 years. Before coming to CRN, he was
a senior investigator and Head of the Natural Products Chemistry Section,
Laboratory of Drug Discovery Research and Development, Developmental Therapeutics
Program, National Cancer Institute.
 Barrett is founder and principal
of Pharmacognosy Consulting Services in Redwood City, CA. She has more than
20 years of experience in natural products research, including postdoctoral
positions at the Royal College of Surgeons and at the William Harvey Research
Institute in London, directing a drug discovery screening program at Shaman
Pharmaceuticals, and serving as Director of Research and Development for
Trout Lake Farm. Barrett is founder and principal
of Pharmacognosy Consulting Services in Redwood City, CA. She has more than
20 years of experience in natural products research, including postdoctoral
positions at the Royal College of Surgeons and at the William Harvey Research
Institute in London, directing a drug discovery screening program at Shaman
Pharmaceuticals, and serving as Director of Research and Development for
Trout Lake Farm.
 Expert reviewers of the CRN report
included Varro E. Tyler, Ph.D., Sc.D., Dean and Distinguished Professor
Emeritus, Purdue University; and Edward M. Croom, Ph.D., School of Pharmacy,
University of Mississippi. Expert reviewers of the CRN report
included Varro E. Tyler, Ph.D., Sc.D., Dean and Distinguished Professor
Emeritus, Purdue University; and Edward M. Croom, Ph.D., School of Pharmacy,
University of Mississippi. 
|
![]()
![]()
![]()
![]()
![]()
![]()
![]()
